The hydroxides They result from the combination of a metal oxide (also called basic oxides) and water. In this way, the composition of hydroxides is given by three elements: oxygen, hydrogen and the metal in question. In combination, the metal always acts as a cation and the hydroxide group (OH–) acts as an anion. For instance:
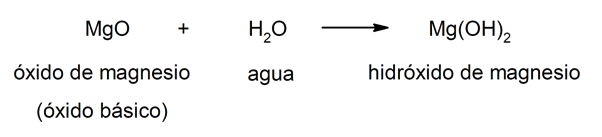
Hydroxides, in general, share a number of characteristics, such as presenting a bitter taste and being caustic. In addition, they are usually slippery to the touch and corrosive. On the other hand, they have some properties of detergents and soaps. Most are soluble in water and react with acids to produce salts.
Some characteristics, on the other hand, are specific to each type of hydroxide, such as that of sodium (NaOH), which absorbs water, and mixed with quicklime (CaO) quickly absorbs carbon dioxide (CO2). For his part, calcium hydroxide (Ca (OH)2) it is obtained in the reaction of calcium oxide with water and also absorbs CO2. Another example is the iron (II) hydroxide (Fe (OH)2), which is a gelatinous compound and is practically insoluble in water.
Hydroxides are also called bases or alkalis, although these terms have been extended also for other substances with alkaline character and that are not hydroxides.
What are hydroxides used for?
The applications of hydroxides also vary between the different cases:
- Sodium hydroxide, for example, is associated with the soap and beauty and body care industries.
- Calcium hydroxide, for its part, has an intermediate role in some processes such as obtaining sodium carbonate (Na2CO3).
- Lithium hydroxide (LiOH) is used in the manufacture of ceramics, while magnesium hydroxide (Mg (OH)2) is used as an antacid or as a laxative.
- Iron hydroxide is used in the plant fertilization process.
Nomenclatures
As with naming other types of chemical compounds, there are different nomenclatures for hydroxides:
- Traditional nomenclature. They are named by putting the word “hydroxide” followed by the name of the metal preceded or terminated in a certain prefix or suffix that depends on the amount of oxidation states it has:
- If the metal has only one oxidation state, its name is given the suffix ‘ico’. For instance: sodium hydroxide / Na (OH).
- If the metal has two oxidation states, its name is given the suffix ‘ico’ when it has the largest and the suffix ‘bear’ when it has the smallest. For instance: ferrous (II) hydroxide / Fe (OH)2 and ferric hydroxide / Fe (OH)3.
- If the metal has three oxidation states, its name is given the prefix ‘hiccup’ and the suffix ‘bear’ when it has the smallest, the suffix ‘bear’ for the one that follows and the suffix ‘ico’ when it has the largest . For instance: hypochromic hydroxide / Cr (OH)2, chromous hydroxide / Cr (OH)3 and chromic hydroxide / Cr (OH)6.
- If the metal has four oxidation states, its name is given the prefix ‘hiccup’ and the suffix ‘bear’ when it has the lowest, the suffix ‘bear’ for the one that follows, the suffix ‘ico’ for the next and the prefix ‘per’ and the suffix ‘ico’ when it has the greater. For instance: hypovanced hydroxide / V (OH)2, vanadic hydroxide / V (OH)3, vanadic hydroxide / V (OH)4 and pervanadic hydroxide / V (OH)5.
- Stock nomenclature. It uses the word hydroxide, but instead of supplementing with a single word, it uses the preposition ‘of’ and then the name of the metal, placing the valence corresponding to each compound in parentheses. For instance: iron (II) hydroxide / Fe (OH)2 and iron (III) hydroxide / Fe (OH)3.
- The systematic nomenclature. They are named by prepending the numerical prefixes to the word ‘hydroxide’. For instance: iron dihydroxide / Fe (OH)2 and iron trihydroxide / Fe (OH)3.
Examples of hydroxides
- Lead (II) hydroxide, Pb (OH)2, lead dihydroxide.
- Platinum (IV) hydroxide, Pt (OH)4, platinum tetrahydroxide.
- Vanadic hydroxide, V (OH)4, vanadium tetrahydroxide.
- Ferrous hydroxide, Fe (OH)2, iron dihydroxide.
- Lead (IV) hydroxide, Pb (OH)4, lead tetrahydroxide.
- Silver hydroxide, AgOH, silver hydroxide.
- Cobalt Hydroxide, Co (OH)2, cobalt dihydroxide.
- Manganese hydroxide, Mn (OH)3, manganese trihydroxide.
- Ferric hydroxide, Fe (OH)3, iron trihydroxide.
- Cupric hydroxide, Cu (OH)2, copper dihydroxide.
- Aluminum hydroxide, Al (OH)3, aluminum trihydroxide.
- Sodium hydroxide, NaOH, sodium hydroxide.
- Strontium hydroxide, Sr (OH)2, strontium dihydroxide.
- Magnesium hydroxide, Mg (OH)2, magnesium dihydroxide.
- Ammonium hydroxide, NH4OH, ammonium hydroxide.
- Cadmium hydroxide, Cd (OH)2, cadmium dihydroxide.
- Vanadic hydroxide, V (OH)3, vanadium trihydroxide.
- Mercuric hydroxide, Hg (OH)2, mercury dihydroxide.
- Cuprous hydroxide, CuOH, copper hydroxide.
- Lithium hydroxide, LiOH, lithium hydroxide.
Hydroxides sometimes have common names given by their more conventional uses: sodium hydroxide is also called caustic soda, potassium hydroxide is called caustic potash, calcium hydroxide is called lime water or slaked lime and magnesium hydroxide is called milk of magnesia .
