In general, when talking about states of the materials we refer to three large groups: solid, liquid and gaseous (although there is also the plasma state).
The gases They are a state of matter in which molecules interact weakly at a certain temperature and pressure. For instance: biogas, carbon monoxide, hydrogen, methane.
At gaseous state, the molecules are not cohesive, so gases do not constitute a consistent body, with a defined shape and volume, as solids do. For this reason, gases are often imperceptible to vision, although some are perceptible to smell.
The gases expand throughout the available space, that is, they adopt the shape and volume of the container that contains them.
State changes:
- The transformation from the solid state to the gaseous state due to a sudden increase in temperature and pressure variation, without previously passing through the liquid state, is called sublimation.
- The transformation from the liquid to the gaseous state by increasing temperature is known as vaporization.
- The transformation from the gaseous state to the liquid by decrease in temperature is called condensation.
Characteristic of gases
- They are very easy to compress.
- They have no definite shape.
- In the gaseous state, the molecules are in permanent motion, the particles colliding with each other and with the walls of the container that contains them.
- These particles move at different speeds according to a certain temperature.
- Movement is faster at elevated temperatures.
- The gravitational and attractive forces are negligible compared to the tendency of the particles that make up gases to move.
Research on gases and air
There are many studies and theoretical contributions in physics and chemistry to analyze the characteristics and behavior of gases.
The air (which almost all living beings need to breathe) must have a certain composition, with a sufficient amount of oxygen. Carbon dioxide is also an important gas in the air, it is needed by plants to carry out the photosynthesis process.
Certain gases should not exceed a certain proportion in the air; In fact, some gases from certain industries are extremely toxic and harmful to health, and can pollute the atmosphere we breathe (the carbon monoxide is an example of them).

Properties of gases
Among the main properties of gases, we find:
- The expansion and the understandability (Gases can be compressed by the action of an external pressure and expand if this pressure is not exerted).
- The diffusion (movement of particles from an area of high concentration to an area of lower concentration) and effusion (When a gas is under pressure in a container and escapes to the outside through an opening).
- The density of a gas is quite low.
The behavior of gases was described through the Gas laws formulated by scientists like Robert Boyle, Jacques Charles, and Gay-Lussac. These physicists related parameters such as volume, pressure and temperature of the gases, which are gathered in the so-called General gas law.
- Boyle’s Law. It establishes that the volume of a gas is inversely proportional to the absolute pressure of the container that contains it at a given temperature and mass of the gas.
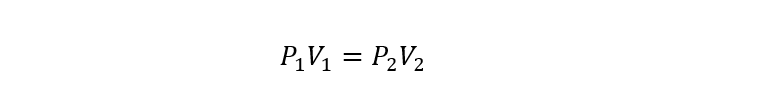
Where:
P1 and P2 are the initial and final pressures of the gas respectively.
V1 and V2 are the initial and final volumes of the gas respectively.
- Law of Jacques. It states that the volume of a gas is proportional to its temperature at a given pressure.
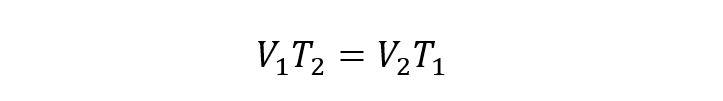
Where:
T1 and T2 are the initial and final temperatures of the gas respectively.
V1 and V2 are the initial and final volumes of the gas respectively.
- Gay-Lussac Law. It states that the pressure of a gas is directly proportional to the temperature when the volume is constant.
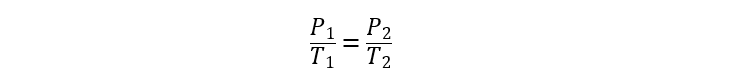
Where:
T1 and T2 are the initial and final temperatures of the gas respectively.
V1 and V2 are the initial and final volumes of the gas respectively.
Examples of matter in the gaseous state

- Emissions coming out of the tailpipe of a moving car.
- The gases used in the refrigeration of refrigerators and air conditioners.
- The clouds in the sky, composed of water vapor.
- Carbon dioxide in carbonated drinks.
- Tear gas, which produces an unpleasant sensation on the human body.
- Gas balloons (filled with helium gas).
- Natural gas used as fuel in the home network.
- Biogas.
- The smoke that is generated when burning any solid.

- Carbon monoxide
- Acetylene
- Hydrogen
- Methane
- Butane
- Ozone
- Oxygen
- Nitrogen
- Hydrogen sulfide gas
- Helium
- Argon
