The hydrocarbons they are organic compounds formed exclusively by a structure of hydrogen and carbon atoms, and which are the basis of all organic chemistry. For instance: methane, hexane, phenol, petroleum. The structure of hydrocarbons can be linear or branched, open or closed. In addition, according to its linear and spatial ordering, and its number of atoms will depend on whether it is one or another substance. They can also have atoms other than carbon and hydrogen in their structure, in which case they are called substituted hydrocarbons.
Hydrocarbons are flammable substances and with a wide capacity for industrial transformation, which is why they constitute the basis of world mining extraction, which allows the development of complex materials, caloric and electrical energy, and lighting, among other possible applications. They are also a considerable source of poisoning, as they often give off vapors that are harmful to health.
Hydrocarbons are classified according to several possible criteria:
According to its structure:
- Acyclic or open chain. In turn, divided into linear or branched.
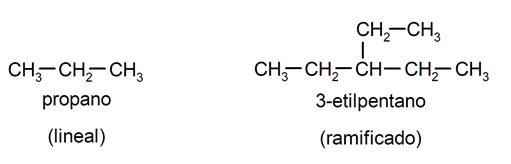
[/su_list]
- Cyclical or closed chain. In turn, divided into monocyclic and polycyclic.
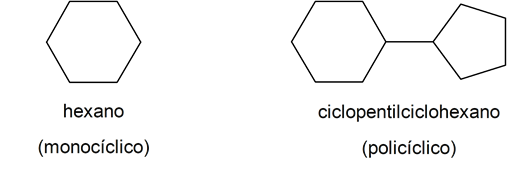
According to the type of bond between its atoms:
- Aromatics. They have at least one aromatic ring, that is to say, with a cyclic structure according to Hückel’s rule. They are derived from Benzene.

- Aliphatic. They lack aromatic ring (not derived from benzene) and in turn are divided into: saturated (single atomic bonds) and unsaturated (at least one double or triple bond).

Examples of hydrocarbons
- Methane (CH4). It is a gas with a repulsive odor, very flammable, present in the atmosphere of the great gaseous planets and in ours it is the product of the decomposition of organic matter or a product of mining activities.
- Ethane (C2H6). It is a highly flammable gas, one of those that constitute natural gas and is capable of producing frostbite in contact with organic tissues.
- Butane (C4H10). It is a colorless and stable fas, widely used as a high pressure (liquid) fuel in the domestic context.
- Propane (C3H8). It is a colorless and odorless gas, endowed with high explosiveness and narcotic properties when exposure to high concentrations occurs.
- Pentane (C5H12). It is an alkane that is normally in a liquid state. It is used as a solvent and as a source of energy, given its high safety and low cost.
- Benzene (C6H6). It is a colorless liquid with a sweet aroma, very flammable and also highly carcinogenic, it is among the industrial products with the highest production today. It is used in the manufacture of rubbers, detergents, pesticides, medicines, plastics, resins and in the refining of petroleum.
- Hexane (C6H14). It is used as a solvent for some paints and adhesives, as well as in obtaining pomace oil. Its use, however, is restricted because it is an addictive neurotoxic.
- Heptane (C7H16). It is a liquid that, at ambient pressure and temperature, is highly flammable and explosive. It is used in the fuel industry as the zero point of octane, and as a working base in pharmaceuticals.
- Octane (C8H18). It is the 100th point on the gasoline octane scale, opposite to heptane, and has a long list of isomers for industrial use.
- 1-Hexene (C6H12). Classified in the industry as a superior olefin and alpha-olefin, it is a colorless liquid essential in obtaining polyethylene and certain aldehydes.
- Ethylene (C2H4). The most widely used organic compound in the world is both a natural plant hormone and an industrial compound necessary for the manufacture of plastic. It is usually obtained from the dehydrogenation of ethane.
- Acetylene (C2H2). It is a colorless gas, lighter than air and highly flammable, which produces a flame capable of reaching 3000 ° C, one of the highest temperatures manageable by man. It is used as a source of lighting and heat in various industries and applications.
- Trichlorethylene (C2HCL3). It is a colorless flammable liquid, with a sweet smell and taste, highly carcinogenic and toxic, capable of interrupting the cardiac, respiratory and hepatic cycles. It is a powerful industrial solvent that does not exist in nature.
- Trinitrotoluene (C7H5N3OR6). Known as TNT, it is a pale yellow, crystalline, highly explosive compound. It does not react with metals or absorb water, so it has a long life and is widely used as part of military and industrial bombs and explosives.
- Phenol (C6H6OR). Also known as carbolic or phenyl acid or phenylhydroxide, it is solid in its pure, crystalline, white or colorless form. It is used to obtain resins, nylon and as a disinfectant or part of various medical preparations.
- Tar. It is a complex mixture of organic compounds whose formula varies according to the nature of its production and its temperature and other variables. It is a liquid, bituminous, viscous and dark substance, with a strong odor and many applications, from psoriasis treatment to road paving.
- Kerosene. It is a common fuel, not very clean and obtained through the distillation of natural petroleum. It is composed of a mixture of hydrocarbons in a transparent and yellowish liquid, insoluble in water, used for lighting and surface cleaning purposes, as well as a pesticide and motor lubricant.
- Gasoline. Obtained from petroleum by direct or fractional distillation, this mixture of hundreds of hydrocarbons is used in internal combustion engines as the cleanest, most efficient and popular fuel known, especially after it was separated from lead in the early 2000s. .
- Petroleum. It is the most important mixture of hydrocarbons known in industrial terms. From oil it is possible to synthesize many other and diverse types of substances. It is produced underground from organic matter accumulated in geological traps and subjected to extremely high pressure. It is of fossil origin, a viscous and dense black liquid, whose world reserves are non-renewable, but which constitutes the main input for the automotive, electrical, chemical and materials industries.


