The alkanes They are a class of hydrocarbons that are made up of a variable number of carbon atoms that are joined together by single bonds, like a skeleton. In turn, each carbon atom is attached to hydrogen atoms, which can eventually be replaced by other atoms or functional groups. For example: chloroform, methane, octane.
The molecular formula of the open linear chain alkanes is CnH2n + 2: C represents carbon, H represents hydrogen and n represents the number of carbon atoms. Alkanes are saturated hydrocarbons, which means they do not have double or triple bonds. To name them, the suffix “-ano” is used, after naming the carbon chain using the prefix corresponding to the number of carbon atoms (et- (2), pro- (3), but- (4), pen- (5), hex- (6), hep- (7)).
Classification of alkanes
Within the alkanes they are usually recognized two large groups: open chain (also called acyclic) and closed chain (or cyclical).
When the open chain compounds They do not present any substitution of the hydrogens that accompany each carbon atom, they are called linear alkanes and are the simplest alkanes. When they present a substitution of any of their hydrogens by one or more carbon chains, they are called branched alkanes. The most common substituents are ethyl groups (CH3CHtwo-) and methyl (CH3-).
On the other hand, there are compounds with a single cycle in the molecule (monocyclic) and others with several (polycyclic). The cyclic alkanes They can be homocyclic (they are formed with the exclusive intervention of carbon atoms) or heterocyclic (in which other atoms participate, for example, oxygen or sulfur).
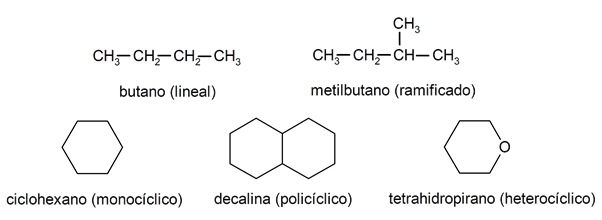
Physical properties of alkanes
In general, the physical properties of alkanes are conditioned by the molecular mass (in turn linked to the length of the carbon chain).
- State of aggregation. Lower carbon alkanes are gaseous at room temperature, those ranging from 5 to 18 carbon atoms are liquid and above 18 atoms are solid (similar to wax).
- Relationship with water. Being less dense than water, alkanes tend to float on it, in general, they are insoluble in water and soluble in organic solvents.
- Boiling and melting point. The boiling and melting point of alkanes depends on their molecular mass, that is, on the length of the carbon chain, although it also often depends on the spatial arrangement of the atoms. Linear and cyclic alkanes have higher boiling points than branched ones.
Chemical properties of alkanes
Some of the main chemical properties of alkanes are:
- They are chemical compounds of very low reactivity, which is why they are also known as “paraffins” (in Latin, parum affinis means “low affinity”).
- They are compounds that have a very high activation energy when they are involved in chemical reactions. The most important reaction they can undergo is combustion and they generate, in the presence of oxygen, heat, carbon dioxide and water.
- They are the basis of an important variety of reactions associated with very important industrial processes, being the most traditional fuels.
- They appear as end products of biological processes such as methanogenic fermentation carried out by some microorganisms.
Examples of alkanes
- Trichloromethane (CHCl3)
- Methane (CH4)
- Ethane (CtwoH6)
- Propane (C3H8)
- Butane (C4H10)
- Octane (C8H18)
- Hexane (C6H14)
- Icosano (CtwentyH42)
- Pentane (C5H12)
- n-heptane (C7H16)
- 2-methylbutane (C5H12)
- 3-chloro-4-n-propylheptane (C10Htwenty-oneCl)
- 1-bromo-2-phenylethane (C8H9Br)
- 3-ethyl-4-methylhexane (C9H18)
- Cuban (C8H8)
- 1-bromopropane (C3H7Br)
- 3-methyl-5-n-propyloctane (C12H26)
- 3,3-dimethyldecane (C12H26)
- Cyclopropane (C3H6)
- 3-ethyl-2,3-dimethylpentane (C9Htwenty)
Alkanes in everyday life
Some of the main uses of alkanes are:
- Chloroform (trichloromethane). Vapors from this substance used to be used as anesthetics, but were later banned because they were found to damage vital organs, such as the liver and kidneys. Trichloromethane is currently used as a solvent and coolant.
- Methane. It is the simplest alkane and is made up of one carbon and four hydrogen atoms. It is a gas that occurs naturally, by the decomposition of different organic substrates, and is the main component of natural gas used to produce electricity. Methane is also used in the chemical industry and is one of the gases that contributes the most to the greenhouse effect.
- Ethane. It is an alkane that is used for the production of ethylene (a compound widely used in industry), to generate energy and in the production of plastics and detergents.
- Propane. It is a gas that is used as an electrical fuel in homes and industries. It is used to heat rooms, heat water and in ovens and kitchens. In addition, it is used as a fuel for vehicles.
- Octane. It is an eight-carbon alkane and is of great importance because it determines the final quality of the gasoline. This quality is measured by the octane number or octane number of the fuel, which takes as a reference a low-detonating one (index 100) and a highly detonating one (index 0).
- Hexane. It is an important solvent and, due to its high toxicity, it is not used frequently.
- Butane. Along with propane (C3H8), make up the so-called liquefied petroleum gases (LPG), which are formed in gas bags during oil extraction processes. Currently, the replacement of gasoline or diesel by LPG is being promoted since it is a more environmentally friendly hydrocarbon.
- Pentane. It is a liquid alkane that is used as a solvent and is part of some gasoline.
- Icosano. It is the twenty-carbon alkane (the prefix ‘ico’ means twenty) and is often used in the manufacture of cosmetics and lubricants.
- Cyclopropane. It is an alkane that was formerly used as an anesthetic, but was replaced by other substances.
- n-heptane. It is an alkane that is taken as a reference for the zero point of the octane scale of gasoline, which would be the least desirable, since it burns explosively. It is obtained from the resin of certain plants and is used as a solvent. It is also used in the oil industry and, to a lesser extent, as a fuel.
- 3-ethyl-2,3-dimethylpentane. It is a compound that is used as a solvent and as a component of gasoline.
- 2-methylbutane. It is a liquid and highly flammable compound, also called isopentane, which is often used as a solvent, for the manufacture of plastics and in the chemical industry.
